Research
Capsule Regulation and Virulence in Cryptococcus Neoformans
The encapsulated fungus Cryptococcus neoformans is responsible for life-threatening disease, particularly in the context of compromised immunity, and current therapy is not adequate. The main virulence factor of C. neoformans is an extensive polysaccharide capsule. The structures of the capsule polysaccharides are known and aspects of capsule construction have been studied, but there is only limited understanding of how the events of capsule synthesis are regulated.
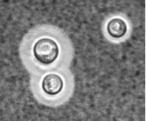


Capsule imaging. Left, ink stain; middle, automated annotation of the image at left, showing cells and capsules (green); right, confocal image with fluorescein-tagged cell wall (green) and antibody tagged capsule (red) from our earlier work.
Our long-term goal is to understand capsule synthesis in detail, so that we can target this process for antifungal therapy. In this application we propose to use a togel online powerful combination of molecular and computational techniques to reconstruct the regulatory network that controls capsule synthesis.
Our first aim is designed to systematically identify genes involved in capsule regulation. Our second aim is designed to computationally reconstruct the capsule regulatory network, enabling quantitative modeling of events in capsule regulation and prioritization of interactions for further study. Our third aim proposes to validate selected regulatory relationships by modulating the expression levels of key transcription factors and to characterize the binding sites of these transcription factors.
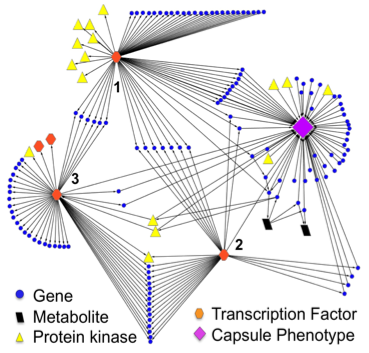
Representation of interactions pertaining to capsule regulation.
Methods include gene deletion, gene expression analysis, microscopy, automated network reconstruction, quantitative modeling, RNA interference, tetracycline regulation, and chromatin immunoprecipitation coupled with sequencing. This range of studies is be enabled by the synergistic efforts of two labs with complementary skill sets, a potent combination that generates significant progress in situs sbobet this important research field. Implementation of this innovative combination of techniques yields greater understanding of cryptococcal biology and pathogenesis, identifing targets for anti-fungal drug discovery, and generating valuable data sets and experimental approaches for future work on this and other eukaryotic pathogens.
Collaborators:
 |
Tamara Doering, Ph.D.Website: www.crypto.wustl.edu |
Relevant Publications:
Gish, S.R., et al. Computational Analysis Reveals a Key Regulator of Cryptococcal Virulence and Determinant of Host Response. MBio 2016;7(2).